Nickel »
PDB 1a5n-1fwb »
1ejt »
Nickel in PDB 1ejt: Crystal Structure of the H219Q Variant of Klebsiella Aerogenes Urease
Enzymatic activity of Crystal Structure of the H219Q Variant of Klebsiella Aerogenes Urease
All present enzymatic activity of Crystal Structure of the H219Q Variant of Klebsiella Aerogenes Urease:
3.5.1.5;
3.5.1.5;
Protein crystallography data
The structure of Crystal Structure of the H219Q Variant of Klebsiella Aerogenes Urease, PDB code: 1ejt
was solved by
M.A.Pearson,
I.S.Park,
R.A.Schaller,
L.O.Michel,
P.A.Karplus,
R.P.Hausinger,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 10.00 / 2.00 |
| Space group | I 21 3 |
| Cell size a, b, c (Å), α, β, γ (°) | 170.800, 170.800, 170.800, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 17.3 / n/a |
Nickel Binding Sites:
The binding sites of Nickel atom in the Crystal Structure of the H219Q Variant of Klebsiella Aerogenes Urease
(pdb code 1ejt). This binding sites where shown within
5.0 Angstroms radius around Nickel atom.
In total 2 binding sites of Nickel where determined in the Crystal Structure of the H219Q Variant of Klebsiella Aerogenes Urease, PDB code: 1ejt:
Jump to Nickel binding site number: 1; 2;
In total 2 binding sites of Nickel where determined in the Crystal Structure of the H219Q Variant of Klebsiella Aerogenes Urease, PDB code: 1ejt:
Jump to Nickel binding site number: 1; 2;
Nickel binding site 1 out of 2 in 1ejt
Go back to
Nickel binding site 1 out
of 2 in the Crystal Structure of the H219Q Variant of Klebsiella Aerogenes Urease
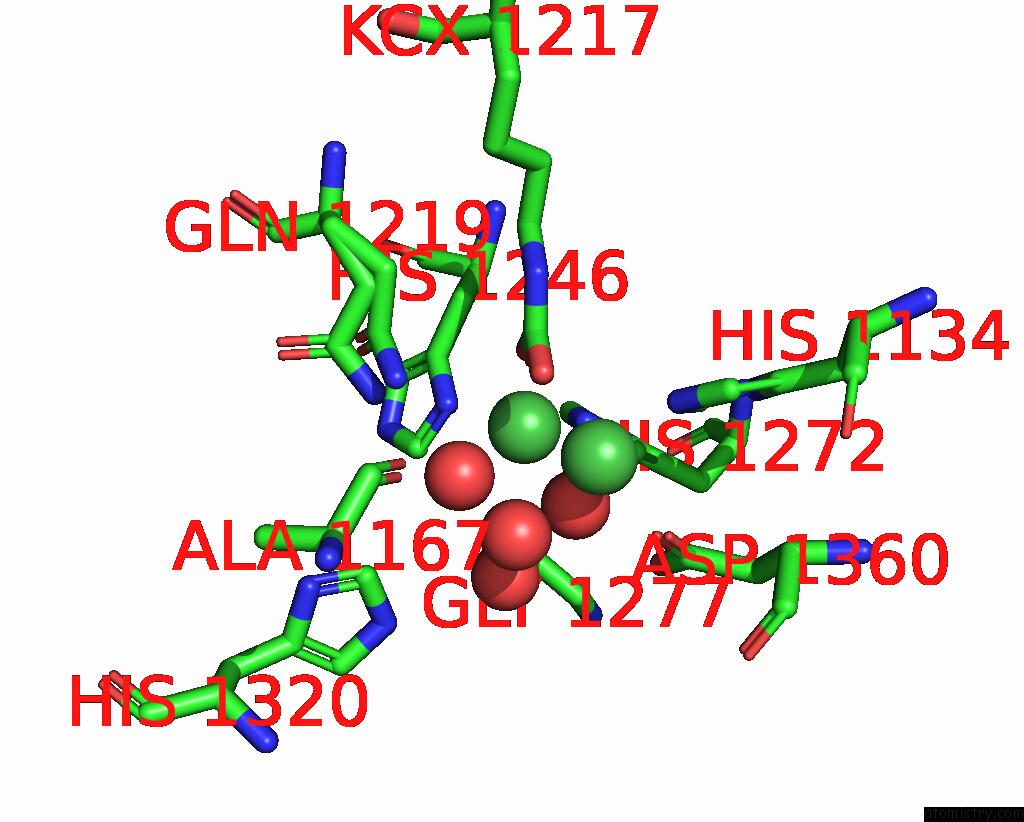
Mono view
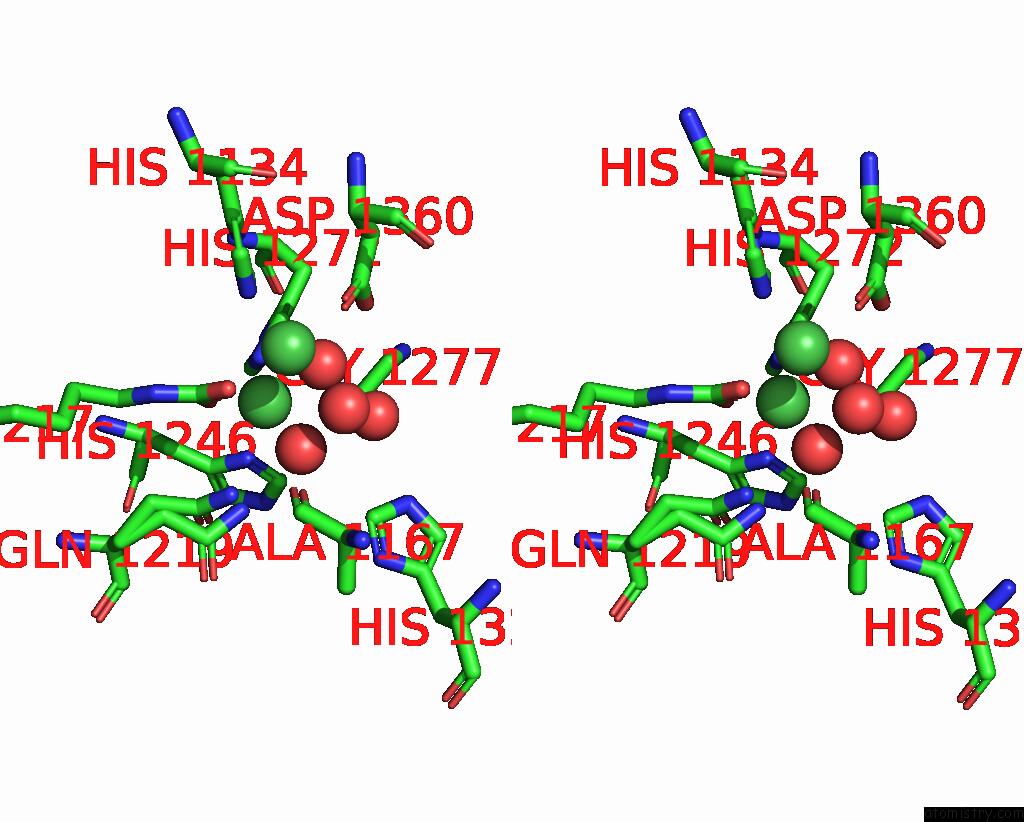
Stereo pair view

Mono view

Stereo pair view
A full contact list of Nickel with other atoms in the Ni binding
site number 1 of Crystal Structure of the H219Q Variant of Klebsiella Aerogenes Urease within 5.0Å range:
|
Nickel binding site 2 out of 2 in 1ejt
Go back to
Nickel binding site 2 out
of 2 in the Crystal Structure of the H219Q Variant of Klebsiella Aerogenes Urease
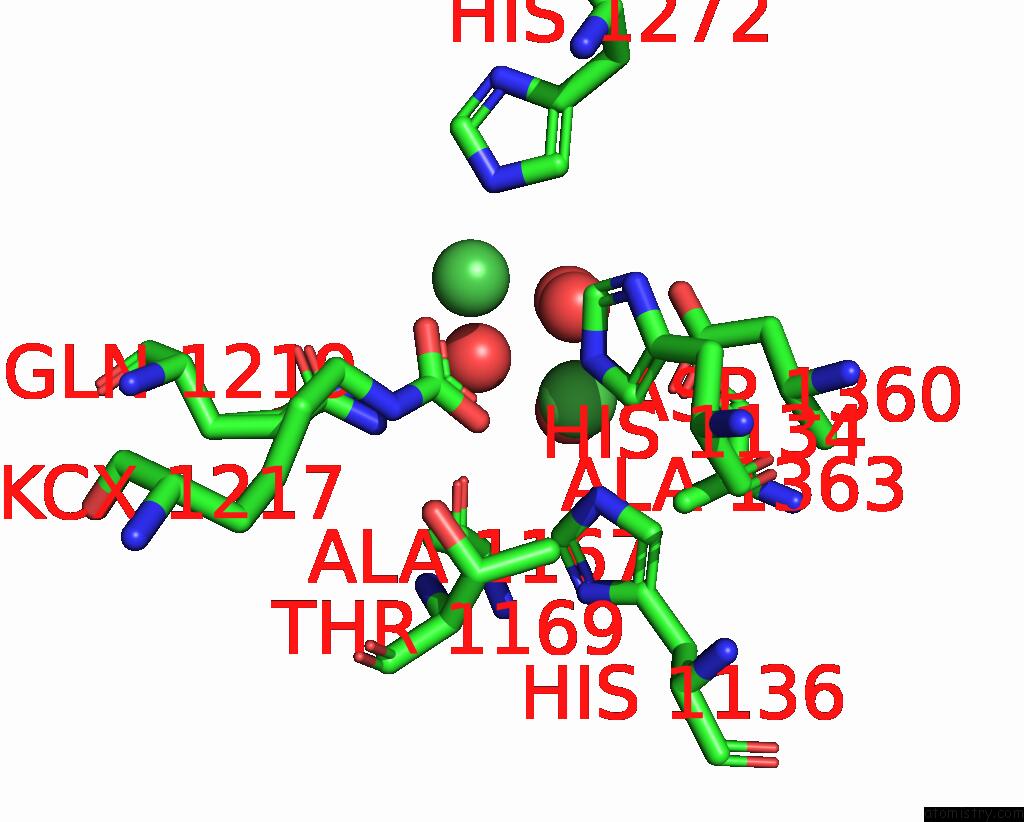
Mono view
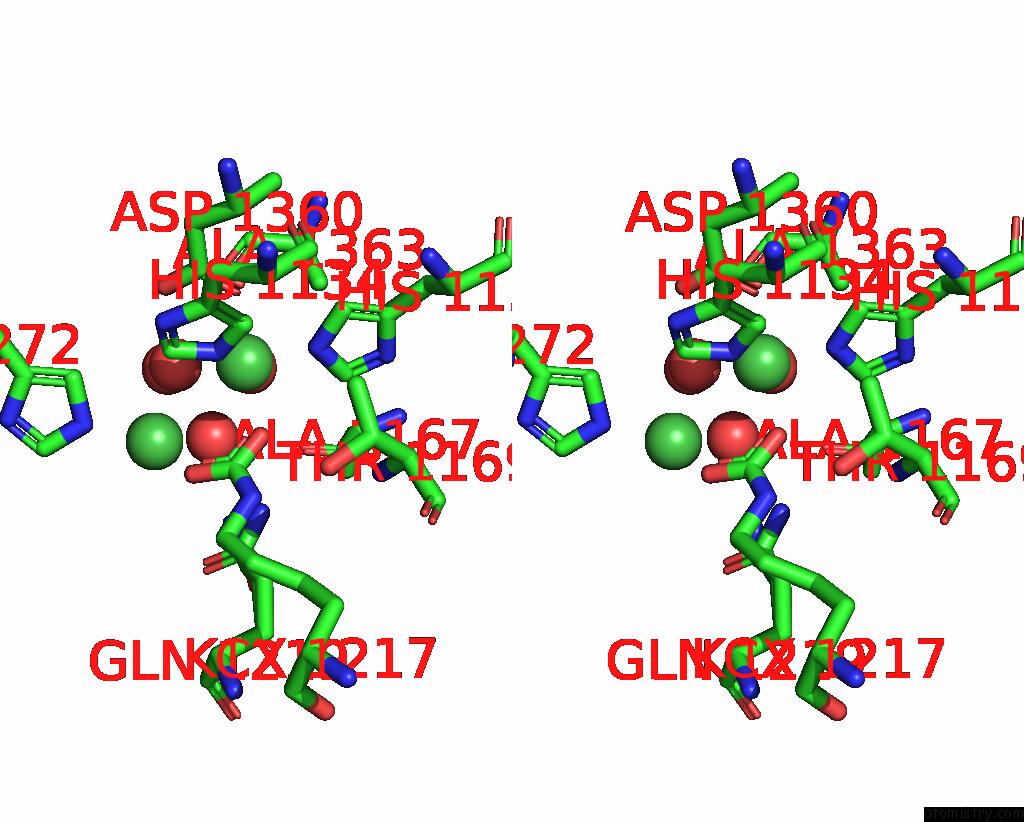
Stereo pair view

Mono view

Stereo pair view
A full contact list of Nickel with other atoms in the Ni binding
site number 2 of Crystal Structure of the H219Q Variant of Klebsiella Aerogenes Urease within 5.0Å range:
|
Reference:
M.A.Pearson,
I.S.Park,
R.A.Schaller,
L.O.Michel,
P.A.Karplus,
R.P.Hausinger.
Kinetic and Structural Characterization of Urease Active Site Variants. Biochemistry V. 39 8575 2000.
ISSN: ISSN 0006-2960
PubMed: 10913264
DOI: 10.1021/BI000613O
Page generated: Mon Aug 18 17:38:55 2025
ISSN: ISSN 0006-2960
PubMed: 10913264
DOI: 10.1021/BI000613O
Last articles
Ni in 5Q4HNi in 5Q4E
Ni in 5Q4G
Ni in 5Q4F
Ni in 5Q4D
Ni in 5Q4B
Ni in 5Q4A
Ni in 5Q4C
Ni in 5Q49
Ni in 5Q47