Nickel »
PDB 1xu1-2c0n »
1zx8 »
Nickel in PDB 1zx8: Crystal Structure of An Atypical Cyclophilin (Peptidylprolyl Cis-Trans Isomerase) (TM1367) From Thermotoga Maritima at 1.90 A Resolution
Protein crystallography data
The structure of Crystal Structure of An Atypical Cyclophilin (Peptidylprolyl Cis-Trans Isomerase) (TM1367) From Thermotoga Maritima at 1.90 A Resolution, PDB code: 1zx8
was solved by
Joint Center For Structural Genomics (Jcsg),
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 29.07 / 1.90 |
| Space group | P 21 21 2 |
| Cell size a, b, c (Å), α, β, γ (°) | 77.083, 132.875, 41.319, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 16.6 / 21 |
Nickel Binding Sites:
The binding sites of Nickel atom in the Crystal Structure of An Atypical Cyclophilin (Peptidylprolyl Cis-Trans Isomerase) (TM1367) From Thermotoga Maritima at 1.90 A Resolution
(pdb code 1zx8). This binding sites where shown within
5.0 Angstroms radius around Nickel atom.
In total only one binding site of Nickel was determined in the Crystal Structure of An Atypical Cyclophilin (Peptidylprolyl Cis-Trans Isomerase) (TM1367) From Thermotoga Maritima at 1.90 A Resolution, PDB code: 1zx8:
In total only one binding site of Nickel was determined in the Crystal Structure of An Atypical Cyclophilin (Peptidylprolyl Cis-Trans Isomerase) (TM1367) From Thermotoga Maritima at 1.90 A Resolution, PDB code: 1zx8:
Nickel binding site 1 out of 1 in 1zx8
Go back to
Nickel binding site 1 out
of 1 in the Crystal Structure of An Atypical Cyclophilin (Peptidylprolyl Cis-Trans Isomerase) (TM1367) From Thermotoga Maritima at 1.90 A Resolution
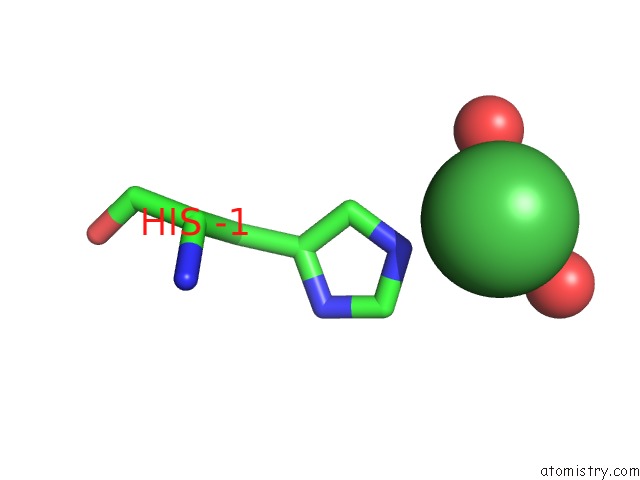
Mono view
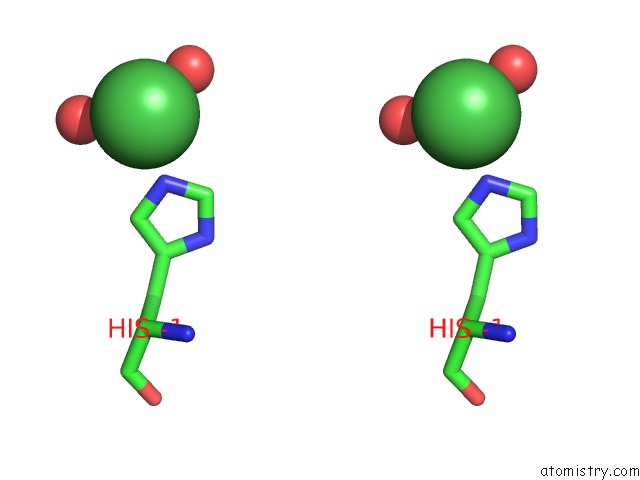
Stereo pair view

Mono view

Stereo pair view
A full contact list of Nickel with other atoms in the Ni binding
site number 1 of Crystal Structure of An Atypical Cyclophilin (Peptidylprolyl Cis-Trans Isomerase) (TM1367) From Thermotoga Maritima at 1.90 A Resolution within 5.0Å range:
|
Reference:
K.K.Jin,
S.S.Krishna,
R.Schwarzenbacher,
D.Mcmullan,
P.Abdubek,
S.Agarwalla,
E.Ambing,
H.Axelrod,
J.M.Canaves,
H.J.Chiu,
A.M.Deacon,
M.Didonato,
M.A.Elsliger,
J.Feuerhelm,
A.Godzik,
C.Grittini,
S.K.Grzechnik,
J.Hale,
E.Hampton,
J.Haugen,
M.Hornsby,
L.Jaroszewski,
H.E.Klock,
M.W.Knuth,
E.Koesema,
A.Kreusch,
P.Kuhn,
S.A.Lesley,
M.D.Miller,
K.Moy,
E.Nigoghossian,
L.Okach,
S.Oommachen,
J.Paulsen,
K.Quijano,
R.Reyes,
C.Rife,
R.C.Stevens,
G.Spraggon,
H.Van Den Bedem,
J.Velasquez,
A.White,
G.Wolf,
G.W.Han,
Q.Xu,
K.O.Hodgson,
J.Wooley,
I.A.Wilson.
Crystal Structure of TM1367 From Thermotoga Maritima at 1.90 A Resolution Reveals An Atypical Member of the Cyclophilin (Peptidylprolyl Isomerase) Fold. Proteins V. 63 1112 2006.
ISSN: ISSN 0887-3585
PubMed: 16544291
DOI: 10.1002/PROT.20894
Page generated: Wed Oct 9 16:35:40 2024
ISSN: ISSN 0887-3585
PubMed: 16544291
DOI: 10.1002/PROT.20894
Last articles
As in 1DVAAs in 1ED4
As in 1DMK
As in 1DMJ
As in 1DMI
As in 1DM8
As in 1DM7
As in 1DM6
As in 1DAN
As in 1D1Y