Nickel »
PDB 1jhj-1q0d »
1oao »
Nickel in PDB 1oao: Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase
Enzymatic activity of Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase
All present enzymatic activity of Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase:
1.2.7.4; 1.2.99.2; 2.3.1.169;
1.2.7.4; 1.2.99.2; 2.3.1.169;
Protein crystallography data
The structure of Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase, PDB code: 1oao
was solved by
C.Darnault,
A.Volbeda,
E.J.Kim,
P.Legrand,
X.Vernede,
P.A.Lindahl,
J.C.Fontecilla-Camps,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 25.00 / 1.90 |
| Space group | C 1 2 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 244.570, 81.890, 167.220, 90.00, 96.19, 90.00 |
| R / Rfree (%) | 14.7 / 17.9 |
Other elements in 1oao:
The structure of Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase also contains other interesting chemical elements:
| Iron | (Fe) | 30 atoms |
| Zinc | (Zn) | 1 atom |
Nickel Binding Sites:
The binding sites of Nickel atom in the Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase
(pdb code 1oao). This binding sites where shown within
5.0 Angstroms radius around Nickel atom.
In total 6 binding sites of Nickel where determined in the Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase, PDB code: 1oao:
Jump to Nickel binding site number: 1; 2; 3; 4; 5; 6;
In total 6 binding sites of Nickel where determined in the Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase, PDB code: 1oao:
Jump to Nickel binding site number: 1; 2; 3; 4; 5; 6;
Nickel binding site 1 out of 6 in 1oao
Go back to
Nickel binding site 1 out
of 6 in the Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase
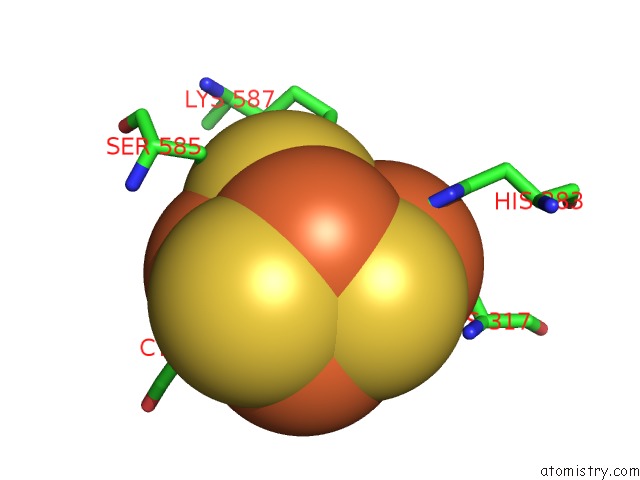
Mono view
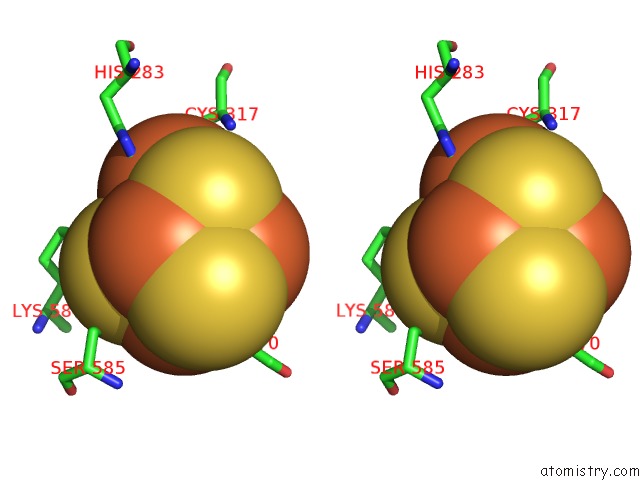
Stereo pair view

Mono view

Stereo pair view
A full contact list of Nickel with other atoms in the Ni binding
site number 1 of Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase within 5.0Å range:
|
Nickel binding site 2 out of 6 in 1oao
Go back to
Nickel binding site 2 out
of 6 in the Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase
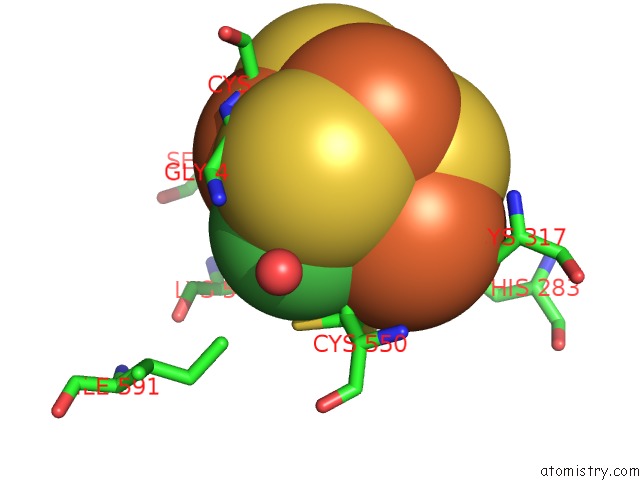
Mono view
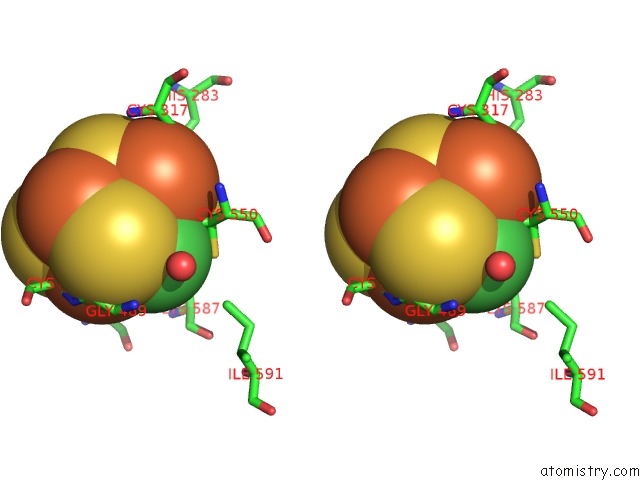
Stereo pair view

Mono view

Stereo pair view
A full contact list of Nickel with other atoms in the Ni binding
site number 2 of Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase within 5.0Å range:
|
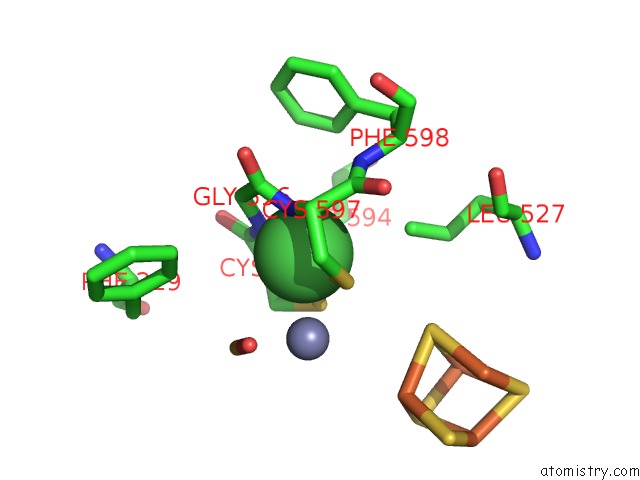
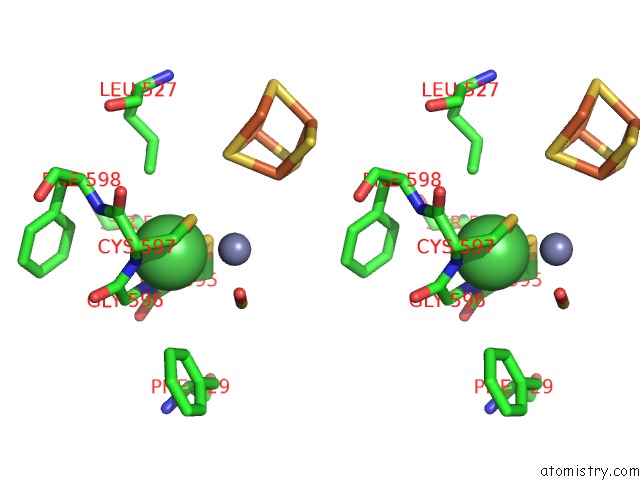
Nickel binding site 3 out of 6 in 1oao
Go back to
Nickel binding site 3 out
of 6 in the Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Nickel with other atoms in the Ni binding
site number 3 of Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase within 5.0Å range:
|
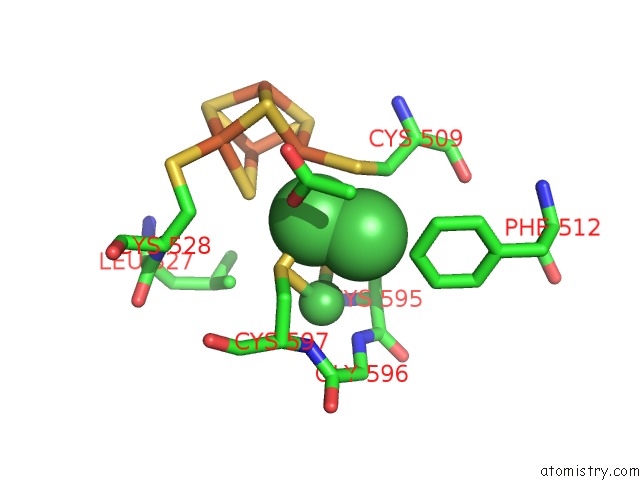
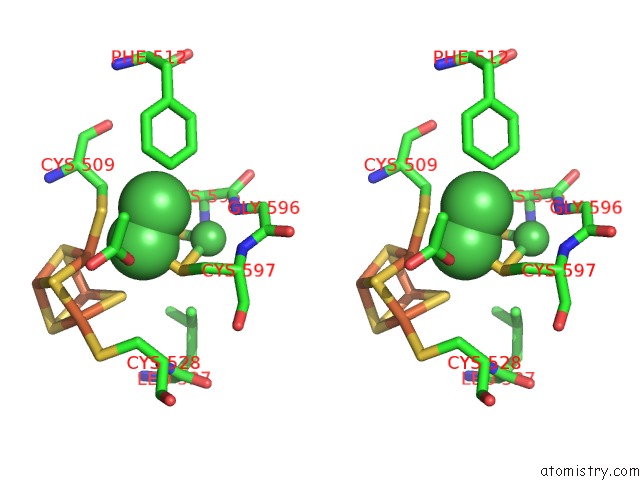
Nickel binding site 4 out of 6 in 1oao
Go back to
Nickel binding site 4 out
of 6 in the Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Nickel with other atoms in the Ni binding
site number 4 of Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase within 5.0Å range:
|
Nickel binding site 5 out of 6 in 1oao
Go back to
Nickel binding site 5 out
of 6 in the Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase
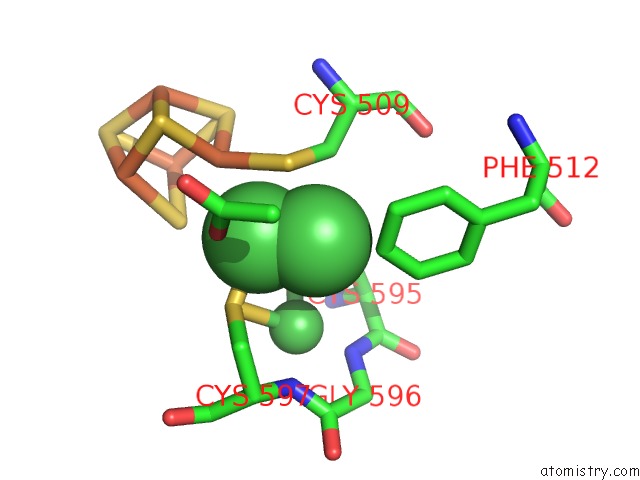
Mono view
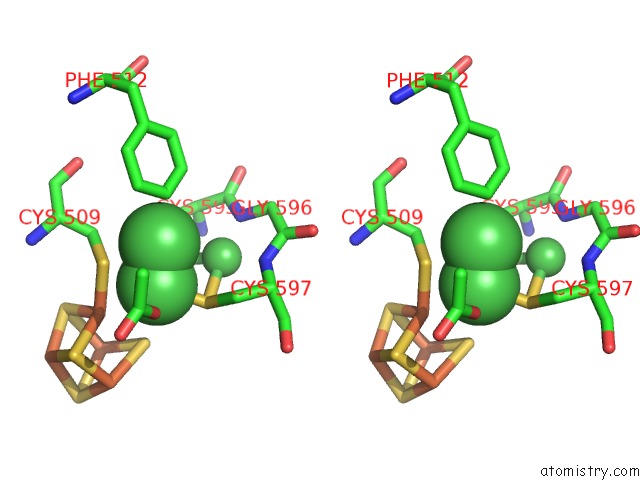
Stereo pair view

Mono view

Stereo pair view
A full contact list of Nickel with other atoms in the Ni binding
site number 5 of Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase within 5.0Å range:
|
Nickel binding site 6 out of 6 in 1oao
Go back to
Nickel binding site 6 out
of 6 in the Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase
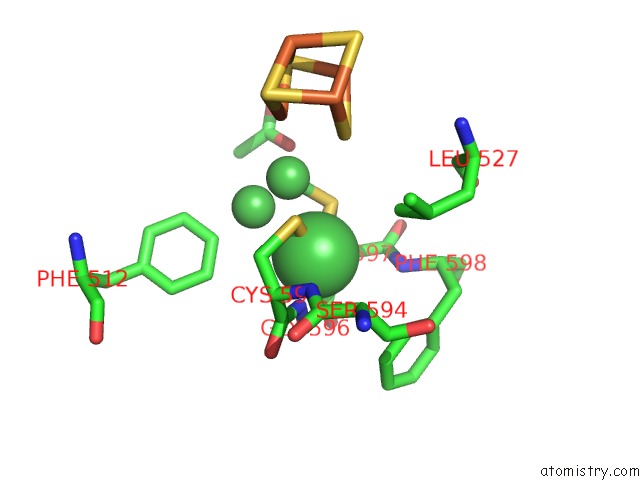
Mono view
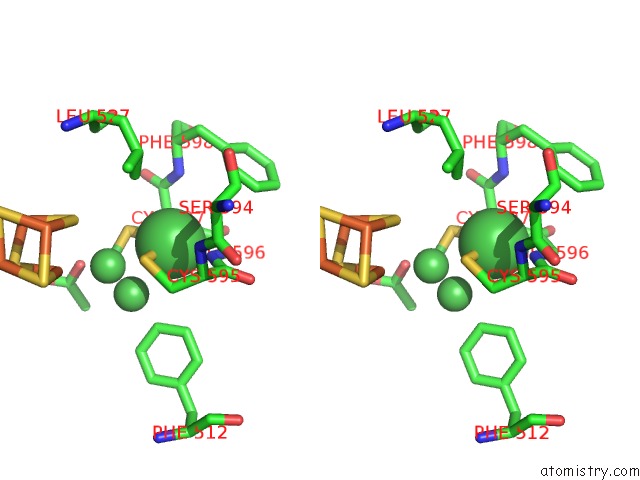
Stereo pair view

Mono view

Stereo pair view
A full contact list of Nickel with other atoms in the Ni binding
site number 6 of Nizn[FE4S4] and Nini[FE4S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase within 5.0Å range:
|
Reference:
C.Darnault,
A.Volbeda,
E.J.Kim,
P.Legrand,
X.Vernede,
P.A.Lindahl,
J.C.Fontecilla-Camps.
Ni-Zn-[FE4-S4] and Ni-Ni-[FE4-S4] Clusters in Closed and Open Alpha Subunits of Acetyl-Coa Synthase/Carbon Monoxide Dehydrogenase Nat.Struct.Biol. V. 10 271 2003.
ISSN: ISSN 1072-8368
PubMed: 12627225
DOI: 10.1038/NSB912
Page generated: Wed Oct 9 15:03:40 2024
ISSN: ISSN 1072-8368
PubMed: 12627225
DOI: 10.1038/NSB912
Last articles
Zn in 9MJ5Zn in 9HNW
Zn in 9G0L
Zn in 9FNE
Zn in 9DZN
Zn in 9E0I
Zn in 9D32
Zn in 9DAK
Zn in 8ZXC
Zn in 8ZUF