Nickel »
PDB 3l1m-3n6n »
3mib »
Nickel in PDB 3mib: Oxidized (CU2+) Peptidylglycine Alpha-Hydroxylating Monooxygenase (Phm) with Bound Nitrite
Enzymatic activity of Oxidized (CU2+) Peptidylglycine Alpha-Hydroxylating Monooxygenase (Phm) with Bound Nitrite
All present enzymatic activity of Oxidized (CU2+) Peptidylglycine Alpha-Hydroxylating Monooxygenase (Phm) with Bound Nitrite:
1.14.17.3;
1.14.17.3;
Protein crystallography data
The structure of Oxidized (CU2+) Peptidylglycine Alpha-Hydroxylating Monooxygenase (Phm) with Bound Nitrite, PDB code: 3mib
was solved by
E.E.Chufan,
B.A.Eipper,
R.E.Mains,
L.M.Amzel,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 52.00 / 2.35 |
| Space group | P 21 21 21 |
| Cell size a, b, c (Å), α, β, γ (°) | 68.612, 68.617, 80.254, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 20 / 24.4 |
Other elements in 3mib:
The structure of Oxidized (CU2+) Peptidylglycine Alpha-Hydroxylating Monooxygenase (Phm) with Bound Nitrite also contains other interesting chemical elements:
| Copper | (Cu) | 2 atoms |
Nickel Binding Sites:
The binding sites of Nickel atom in the Oxidized (CU2+) Peptidylglycine Alpha-Hydroxylating Monooxygenase (Phm) with Bound Nitrite
(pdb code 3mib). This binding sites where shown within
5.0 Angstroms radius around Nickel atom.
In total only one binding site of Nickel was determined in the Oxidized (CU2+) Peptidylglycine Alpha-Hydroxylating Monooxygenase (Phm) with Bound Nitrite, PDB code: 3mib:
In total only one binding site of Nickel was determined in the Oxidized (CU2+) Peptidylglycine Alpha-Hydroxylating Monooxygenase (Phm) with Bound Nitrite, PDB code: 3mib:
Nickel binding site 1 out of 1 in 3mib
Go back to
Nickel binding site 1 out
of 1 in the Oxidized (CU2+) Peptidylglycine Alpha-Hydroxylating Monooxygenase (Phm) with Bound Nitrite
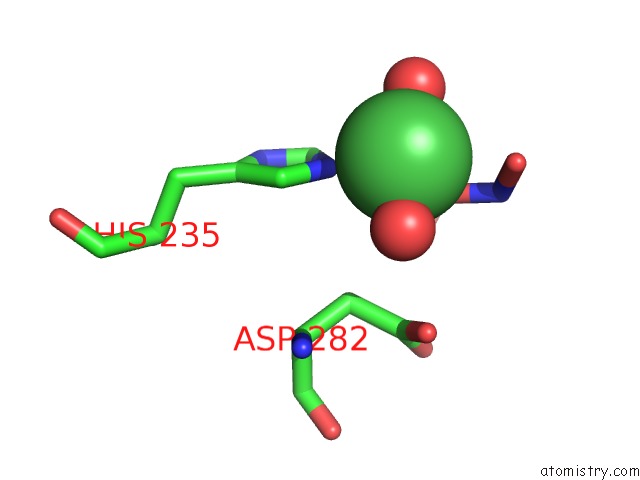
Mono view
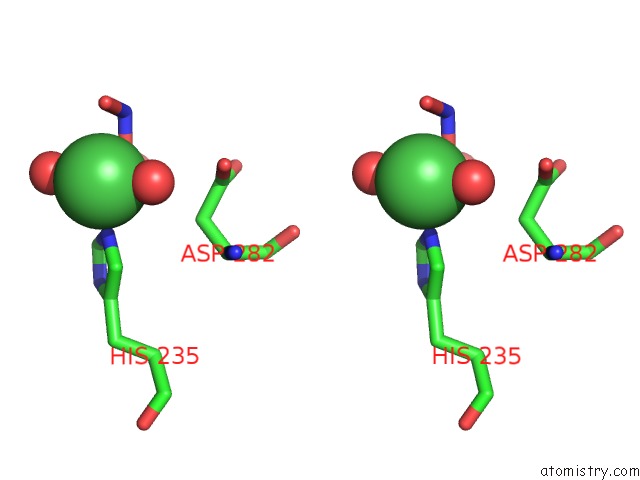
Stereo pair view

Mono view

Stereo pair view
A full contact list of Nickel with other atoms in the Ni binding
site number 1 of Oxidized (CU2+) Peptidylglycine Alpha-Hydroxylating Monooxygenase (Phm) with Bound Nitrite within 5.0Å range:
|
Reference:
E.E.Chufan,
S.T.Prigge,
X.Siebert,
B.A.Eipper,
R.E.Mains,
L.M.Amzel.
Differential Reactivity Between the Two Copper Sites of Peptidylglycine Alpha-Hydroxylating Monooxygenase (Phm) J.Am.Chem.Soc. V. 132 15565 2010.
ISSN: ISSN 0002-7863
PubMed: 20958070
DOI: 10.1021/JA103117R
Page generated: Mon Aug 18 18:47:00 2025
ISSN: ISSN 0002-7863
PubMed: 20958070
DOI: 10.1021/JA103117R
Last articles
Ni in 5WK0Ni in 5WAY
Ni in 5WJA
Ni in 5VMP
Ni in 5VGI
Ni in 5VB0
Ni in 5VP3
Ni in 5UDW
Ni in 5VFB
Ni in 5V9T